Document Type : Original Article
Authors
1 Department of Food Hygiene, Faculty of Veterinary Medicine, Ferdowsi University of Mashhad, Mashhad, Iran
2 Department of Clinical Sciences, Faculty of Veterinary Medicine, Ferdowsi University of Mashhad, Mashhad, Iran
Abstract
The purpose of this study was to elucidate some factors affecting the growth of Salmonella typhimurium. These factors included Carum copticum essential oil (0%, 0.015%, 0.03% and 0.06%), temperature (25 ˚C and 35 ˚C), pH (5, 6 and 7) and inoculum size (103 and 105
CFU mL-1). Brain heart infusion broth was used as the growth medium. There were 48 treatment combinations and the experiment was carried out in triplicate. Growth was monitored by visible turbidity over a 30 days period. A parametric survival model based on the log-normal distribution was used to estimate the most influential factors on the time to detection of Salmonella growth. According to our results, the selected factors significantly affected the growth of Salmonella typhimurium. Furthermore, the final graph demonstrated good agreement between the values predicted by predictive model and the results which were observed in this study. So that a parametric survival model can be a useful and practical tool to predict how the parameters will influence the bacterial growth.
Keywords
Main Subjects
Introduction
Salmonella species have been considered as one of the most important foodborne pathogens all around the world.1,2 Animals are the principal reservoir of this pathogen.3 Salmonella enterica serovar typhimurium and Salmonella enterica serovar enteritidis are the most frequently isolated serovars from foodborne outbreaks throughout the world.4
The use of natural antimicrobial compounds is of utmost interest in the food industry.5 In this regard, the antimicrobial properties of plant essential oil (EO) are investigated in different studies.6,7The combination of different preservation methods have been already described as hurdle technology concept in food safety by Leistner.8
Microbial responses to environmental conditions are reproducible and this phenomenon is the premise of predictive microbiology. We can predict the behavior of the same organism at the similar environmental condition based on previous observations of microbial growth.9,10
It is important to determine the minimum concentration of EO that can inhibit the growth of pathogens without affecting the organoleptic properties of the food.11 Among traditional potential herbs used as spice in day to day life, Ajwain (Trachyspermum ammi L. or Carum copticum Benth. and Hook) belonging to Apiaceae family, is widely used for curing various diseases in both humans and animals. In Iran, the fruits of C. copticum is known as Zenyan. Trachyspermum ammi is a grassy, annual plant which grows in the east of India, Iran and Egypt with a white flower and small, brownish seeds.12
Carum copticum fruit, due to favorite its taste, is commonly used in Iran as common spice and food preservative, especially for traditional meat products.13-16
Overall, according to many prior studies, the C. copticum EO exhibits potent antimicrobial and antioxidant activities supporting its potential use for perishable and high fatty foods. 17-19
The objective of this study was to assess the effects of four different factors: inoculum size, temperature, pH and different concentrations of C. copticum EO on the growth/no growth domain of S. typhimurium.
Materials and Methods
Experimental design. To assess the combined effects of C. copticum EO, pH (adjusted by normal solution of HCl), inoculum size and temperature on growth initiation of S. typhimurium, the experiment was arranged in a factorial design. This design included four concentrations of the EO (0, 0.015, 0.03 and 0.06%), three levels of pH (5, 6 and 7), two inoculum levels (103 and 105CFU mL1), two incubation temperatures (25 ˚C and 35 ˚C) and repeated observations (daily) for growth initiation in brain heart infusion (BHI, Merck, Darmstadt, Germany) broth for up to 30 days. S. typhimurium ATCC 14028 (Mast International Ltd., Berkshire, UK) was used as the test organism in this study.
Preparation of inocula. The reference bacterium was plated on Mac Conkey-Agar plates (Merck) and incubated at 37 ˚C for 24 hr. Inoculums were prepared by transferring a loop full of the bacterial colonies to isotonic saline solution in a sterile cuvette to adjust the absorbance of 0.133 at 600 nm using a spectrophotometer (Model 6105; Jenway, Essex, UK). This adjustment gave a cell concentration of 1.50 × 108 CFU mL-1 (equal to No: 0.50, McFarland standards). Tenfold serial dilution was prepared from the adjusted suspension.
Plant material. The pure C. copticum EO was extracted by steam distillation at the Nader-Co® (Agro-Industry Co., Mashhad, Iran).
Gas chromatography-mass spectrometry (GC-MS) analyses. The GC-MS analyses were carried out using an Agilent HP-6890 gas chromatograph (Agilent Technologies, Palo Alto, USA) with HP-5MS capillary column (30 m × 0.25 mm, 0.25 µm film thickness) equipped with an HP 5973 mass spectrometer (Agilent Technologies) with electron impact ionization (70 eV). The oven temperature was maintained at 50 ˚C for 5 min initially, raised at the rate of 3˚C per min to 240 ˚C, finally raised to 290 ˚C at 15 ˚C per min and then held isothermal for 3 min. Helium was used as carrier gas at a flow rate of 0.80 mL per min, samples of 1.00 µL were injected manually in the split less mode. Peak area percent were used for obtaining quantitative data. Retention indices were calculated for all components using a homologous series of n-alkanes injected in conditions equal to samples ones. The components of the oil were identified by comparing their retention indices relative to (C8 to C22) n-alkanes with those of literature or with those of authentic compounds available in our laboratory and confirmed by matching their mass spectra with those of a computer library of the GC-MS data system and other published mass spectra.20
Determinationofminimuminhibitoryconcentration (MIC). To evaluate MIC of the EO, standard tube dilution (macrobroth dilution) technique was used.21 Briefly, the experiment was performed by preparing twofold serial dilutions of EO in BHI broth. The EO was emulsified into BHI broth medium to get a concentration of 1% (100 µL 10 mL1) using (DMSO, Merck, Hohenbrunn, Germany) 5% (v/v), then serially diluted to achieve 50, 25, 12.5, 6.25, 3.125, 1.56, 0.78, 0.40, 0.20 and 0.10 µL 10 mL-1 (from 1% to 0.001%), respectively. Amount of 106 CFU mL1 of test organism was transferred to the test tubes which had different concentrations of EO. The negative control tubes contained different concentrations of EO without inoculation of the bacteria. Both sets of tubes (tests and controls) were incubated at 35 ˚C for 24 hr. The tube with the lowest concentration of EO which had no visible bacterial growth (no turbidity in the tube) was determined as MIC. In this study four different concentrations of EO lower than MIC were selected as test concentrations.
Performingtheexperiment. To produce and maintain a stable oil–water emulsion in broth substrate during the period of study (30 days), we used the method explained by Mann and Markhamwith some modifications.22 Briefly, 5% (v/v) DMSO (Merck) was added as an emulsifier and 0.05% (w/v) agar-agar (Merck) as stabilizer was introduced to the broth substrate. The same amounts of DMSO and agar agar were also added to the combinations with no EO (0.00%) to consider any likely effects on the growth of test organism. The final volume was brought to 100 mL with additional distilled water. After preparation of BHI broth, pH was adjusted using a pH meter (Jenway Ltd.) and normal solution of HCl as acidulant. The content of each flask was autoclaved at 121 ˚C for 15 min. After cooling, the pH of each combination was measured and adjusted again to the considered pH using 1 N filter sterilized HCl (or NaOH; Merck). Then, filter sterilized EO was added in different amounts to satisfy the experimental design. The content of flask containing sterile BHI broth was dispensed in portions of 3 mL into sterile caped tubes (16 × 100 mm; Becton Dickinson, Durham, USA). The tubes were inoculated with S. typhimurium (105 and 103 CFU mL-1). For each combination, the inoculated tubes were incubated at 35 ˚C and 25 ˚C for up to 30 days. During the period of incubation, all tubes were observed for visible growth (turbidity) daily. The number of tubes (combinations) showing growth at a particular observation was recorded. For each combination, a negative control (uninoculated tube) was used. All experiments were conducted in independent triplicate. This design (4 × 3 × 2 × 2 equal to 48 combinations) with three replicates, to prepare 144 treatment tubes, has been evaluated in this study.
Statisticalanalysis. The outcome variable for consideration in this study was the time to visible growth. Since some combinations did not grow until the end of the study, standard regression methods were considered inappropriate. Instead, event-time (survival) analysis was employed which was able to use all experimental data irrespective of whether or not growth occurred.
Parametric survival model based on accelerated failure time (AFT) approach16,23 was used to quantify the effect of each of prescribed explanatory variables on detection time of bacterial growth. The general form of AFT model is:
log(t) = (α+β1x1i + … + βmxmi)+log (τ)
where, log (t) is the natural logarithm of the time to failure (growth), α is an intercept term, β1x1i + … + βmxmi are linear combinations of the m explanatory variables and their regression coefficients and log (τ) is the error term. Using this approach, the AFT coefficients represent the expected change in log (t) for changes in the predictor levels.
In present study, we evaluated the exponential, Weibull, log-normal and log-logistic distributions that can be interpreted in the AFT metric. To evaluate the fitness of candidate distribution to the current data, the mean square error (MSE) was compared. The smaller MSE value indicates a better fit.
MSE = Σ (Predicted – Observed)2 / (n – p)
where, n is the number of observations and p is the number of parameters to be estimated.
To select those explanatory variables that best explained time to detection, a backward stepwise approach was used. Explanatory variables that were not statistically significant were removed from the model one at a time, beginning with the least significant, until the estimated regression coefficients for all retained variables were significant at an alpha level less than 0.05.
Results
Chemical composition of C. copticum EO. The components of oil were determined by GC-MS analysis. The constituents of C. copticum EO, accompanied by retention time and percent, are listed in Table 1. The GC-MS analysis resulted in identification of 16 components representing 98.88% of total oil. The main constituents were thymol (57.18%), ρ-cymene (22.55%), γ-terpinene (13.07%) and trans-anethole (1.70%).
MIC assay. In vitro antibacterial activities of C. copticum EO were assessed by standard tube dilution technique against S. typhimurium. Antibacterial activity was expressed as MIC value. In this study, the MIC of the EO was 1.25 μL mL-1. Then, four different concentrations of EO lower than MIC were selected as test concentrations.
Table 1. Essential oil composition of Carum copticum identified by gas chromatography-mass spectrometry.
|
Phytochemicals |
Percent |
Retention index (RI) |
|
α -Pinene |
0.29 |
11.35 |
|
β -Pinene |
0.43 |
13.45 |
|
β -Myrcene |
0.34 |
14.28 |
|
α-Phellandrene |
0.06 |
14.89 |
|
α -Terpinene |
0.31 |
15.54 |
|
ρ-Cymene |
22.55 |
16.21 |
|
β-Phellandrene |
0.54 |
16.29 |
|
γ -Terpinene |
13.07 |
17.93 |
|
α -Terpinolene |
0.09 |
19.18 |
|
α -Terpineol |
0.15 |
24.92 |
|
L-Carvone |
0.90 |
27.97 |
|
Trans-Anethole |
1.70 |
28.68 |
|
Thymol |
57.18 |
29.73 |
|
Carvacrol |
0.52 |
29.84 |
|
3-Dodecen-1-Al |
0.16 |
36.51 |
|
Apiol |
0.56 |
42.73 |
|
Total |
98.88 |
- |
Description of growth/no growth. About 85.40% of the treatment tubes (123 out of 144) showed growth during the study period. On the basis of MSE value, the log-normal model provided the best fit to the data. The MSE value of the log-normal model was 62.06, while the MSE values were 119.62, 63.03 and 190.21 for weibull, log-logistic and exponential models, respectively.
Evaluation of time to detect the bacterial growth. Median time to detection of bacterial growth was 3 days. Kaplan-Meier survival curve for explanatory variables is presented in Figure 1. The final model equation as below:
TTD=e-0.43+0.72T25+0.36IL1000+0.01EO0.015+2.46EO0.06+1.14PH5+0.17pH6
where, in this equation TTD is time to detection, e is a mathematical constant approximately equal to 2.718281828, T is temperature, IL is inoculum size and EO is essential oil.
This model showed that all explanatory variables had significant association with TTD (Table 2). On average, TTD for treatment combinations with 0.03% and 0.06% of C. copticum EO was 2.59 and 11.70 times greater than those without it, respectively but 0.015% of EO didn’t influence the TTD significantly. The TTD for those combinations with pH levels of 6 and 5 was 1.18 and 3.13 times greater than those with pH level of 7, respectively.
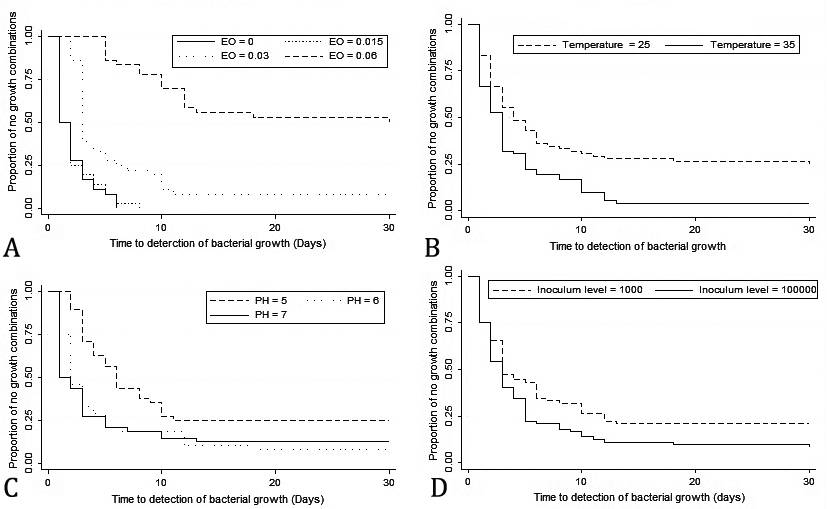
Fig. 1. Kaplan-meier survival curves showing the proportion of no growth combinations; A) concentrations of essential oil, B) temperature, C) pH, and D) inoculum size.
Also, this period for combinations with inoculum size of 103 CFU mL-1 was 1.44 times greater than combinations with inoculum size of 105 CFU mL-1. Furthermore, this period for combinations with incubation temperature of 25 ˚C was 2.06 times greater than combinations with incubation temperature of 35 ˚C.
Observed and predicted TTDs of bacterial growth for each combination are presented in Figure 2.

Fig. 2. Observed and predicted days needed for growth initiation of S. typhimurium according to the log-normal model.
Table 2. Accelerated failure time model of factors influencing time to detection of bacterial growth.
|
Variables |
Measure |
β (SE) |
p-value |
TTD ratio (95% CI) |
|
Intercept |
- |
-0.43 (0.09) |
< 0.01 |
- |
|
Essential oil (%) |
0 |
0 |
- |
1 |
|
0.01 |
0.01 (0.09) |
0.90 |
1.01 (0.85-1.20) |
|
|
0.03 |
0.95 (0.09) |
< 0.01 |
2.59 (2.17-3.10) |
|
|
0.06 |
2.45 (0.09) |
< 0.01 |
11.70 (9.65-14.19) |
|
|
pH |
7 |
0 |
- |
1 |
|
6 |
0.17 (0.07) |
0.03 |
1.18 (1.02-1.38) |
|
|
5 |
1.14 (0.08) |
< 0.01 |
3.13 (2.66-3.68) |
|
|
Inoculum size (CFU mL-1) |
105 |
0 |
- |
1 |
|
103 |
0.36 (0.06) |
< 0.01 |
1.44 (1.26-1.64) |
|
|
Temperature (˚C) |
35 |
0 |
- |
1 |
|
25 |
0.72 (0.06) |
< 0.01 |
2.06 (1.81-2.35) |
|
|
Sigma |
- |
0.38 (0.02) |
- |
- |
β: Regression coefficient; SE: Standard error; CI: Confidence interval; TTD: Time to detection.
Discussion
In the present study, we determined different combinations which can decrease/stop S. typhimurium growth initiation. Using predictive microbiology, we designed a model which can predict growth/no growth of S. typhimurium in different conditions.
An important characteristic of EOs and their components is their hydrophobicity, which enable them to partition the lipids of the bacterial cell membrane and mitochondria, disturbing the cell structures and rendering them more permeable. Extensive leakage from bacterial cells or the exit of critical molecules and ions will lead to death.24,25
In general, EOs containing phenolic compounds, e.g. thymol, carvacrol, terpinene and p-cymene, are widely reported to possess high levels of antibacterial activity and may have applications in controlling pathogens in food.11,26,27 In the present study, the phytochemical components of the EO of C. copticum have been analyzed by GC-MS and thymol (57.18%), p-cymene (22.55%) and γ-terpinene (13.07%) were the major components of the EO. In some studies, amount of thymol has been reported greater than which we analyzed,28 while other reports indicated that thymol is the major constituent of C. copticum EO but mostly less than 50.00% of all components.29-31 It is believed that differences in essential oil profile in a certain plant are resulted from various cultivation situations and locations, time of cultivation and also different extracting method.32
Antimicrobial activity of C. copticum which could be attributed to high content of compounds such as thymol, has been reported by large numbers of studies.15,16,30
The inhibitory effects of EOs increased with increasing their concentration.33 In the present study, similar results were obtained and TTD for combinations with 0.03% and 0.06% of C. copticumEO was 2.59 and 11.70 times greater than those without EO, respectively but 0.015% of EO didn’t impact the TTD significantly.
Singh et al. suggested that the use of EOs in conjunction with other preservation techniques such as chemical preservatives or low temperature could develop a synergistic alternative to the current method.34 Tassou et al. showed a significant extension of detection time of S. aureus and S. enteritidis growth in a nutrient broth medium at a given concentration of mint (Laminaceae) EO, by decreasing the storage temperature from 35 to 10 ˚C. It may be due to the lower metabolic activity at lower temperature.35 Based on our results, decreasing incubation temperature had also a significant effect on growth initiation of inoculated bacteria. Initiation of visible growth period for combinations with incubation temperature of 25 ˚C was 2.06 times greater than those with the same parameters, but incubation temperature of 35 ˚C.
The combined bacteriostatic effect of both low pH and low temperature has been proved to be very effective against Salmonella species.36 The effect of pH can be attributed either to the direct effect of pH or to the better dissolving of the EO in the lipid phase of the bacterial membrane at the lower pH.37 Lowering the pH of a food product or beverage has been used as a preserving method.38 According to our results, decreasing the pH of broth medium had a significant effect on growth initiation of inoculated bacteria.
There are some reports about the effect of inoculum size on bacterial growth.39 It has also been reported that by higher inoculum size, growth can be initiated at the lower pH.40,41 In this regard, Skandamis et al. modeled the effect of inoculum size on growth interface of E. coli O157:H7. They found that at high inoculum size, the intensity of additional environmental inhibitory factors should be higher, which could be explained by the fact that at high inoculum size, there will be more probability of finding cells in the proper physiological state to initiate growth.42 According to our results, S. typhimurium growth was significantly influenced by inoculum size significantly. Initiation of visible growth period for combinations with inoculum size of 103 CFU mL-1 was 1.44 times greater than those with the same parameters but with inoculum size of 105.
Combined data from growth response of a given bacteria in different environmental conditions could be explained in a model. The model offers a cost-effective approach to understand the microbial growth response in foods and forms a data-base against which other controlling factors could be evaluated.43
Based on our results, EO, pH, temperature and inoculum size had significant effects on TTD of S. typhimurium. Furthermore, the log-normal model which has used in this study can be a useful and practical tool to predict how the parameters will influence the bacterial growth. The predicted values may not match with whatever would occur in any special food system. This means that the model must be validated before use in any interested food.
Acknowledgements
The authors express their sincere appreciation and thanks for the financial support received from Faculty of Veterinary Medicine, Ferdowsi University of Mashhad for conducting this research (Grant no: 17485/3). We also thank Mrs. Samira Khajenasiri for her technical assistance.
- Gillespie BE, Mathew AG, Draughon FA, et al. Detection of Salmonella enterica somatic groups C1 and E1 by PCR-enzyme-linked immunosorbent assay. J Food Prot 2003; 66: 2367-2370.
- Malorny B, Hoorfar J, Bunge C, et al. Multicenter validation of the analytic accuracy of Salmonella PCR: toward an international standard. Appl Environ Microbiol 2003; 69: 290-296.
- Winfield MD, Groisman EA. Role of nonhost environments in the lifestyles of Salmonella and Escherichia coli. Appl Environ Microbiol 2003; 69: 3687-3694.
- Herikstad H, Motarjemi Y, Tauxe RV. Salmonella surveillance: A global survey of public health serotyping. Epidemiol Infect 2002; 129: 1-8.
- Kobilinsky A, Nazer AI, Dubois-Brissonnet F. Modeling the inhibition of Salmonella typhimurium growth by combination of food antimicrobials. Int J Food Microbiol 2007; 115: 95-109.
- Cao L, Yong Si J, Liu Y, et al. Essential oil composition, antimicrobial and antioxidant properties of Moslachinensis Maxim. Food Chem 2009; 115: 801-805.
- Hanamanthagouda MS, Kakkalameli SB, Naik PM, et al. Essential oils of Lavandula bipinnata and their antimicrobial activities, short communication. Food Chem 2010; 118: 836-839.
- Gould GW, Abee T, Granum PE, et al. Physiology of food poisoning microorganisms and the major problems in food poisoning control. Int J Food Microbiol 1995; 28(2): 121-128.
- Ross T, McMeekin TA. Predictive microbiology. Int J Food Microbiol 1994; 23: 241-264.
- McMeekin TA, Olley J, Ratkowsky DA, et al. Predictive microbiology: Towards the interface and beyond. Int J Food Microbiol 2002; 73: 395-407.
- Burt S. Essential oils: Their antibacterial properties and potential applications in foods-a review. Int J Food Microbiol 2004; 94: 223-253.
- Qureshi A, Eswar Kumar K. Phytochemical constituents and pharmacological activities of Trachyspermum ammi. Plant Archives 2010; 10(2): 955-959.
- Hashemi SMB, Gavahian M, Mousavi Khaneghah A, et al. Carum copticum essential oils as natural antioxidant in dressing. J Hyg Eng Des 2013; 4: 93-97.
- Hashemi SMB, Mousavi Khaneghah A, Akbarirad H. Effects of ultrasound treatment and zenyan essential oil on lipid oxidation of blended vegetable oil. Int Food Res J 2015; 22(5): 1918-1923.
- Mahboubi M, Kazempour N. Chemical composition and antimicrobial activity of Satureja hortensis and Trachyspermum copticum essential oil. Int J Food Microbiol 2011; 3: 194-200.
- Oskuee RK, Behravan J, Ramezani M. Chemical composition, antimicrobial activity and antiviral activity of essential oil of Carum copticum from Iran. Avicenna J Phytomed 2011; 1: 83-90.
- Hassanshahian M, Bayat Z, Saeidi S, et al. Antimicrobial activity of Trachyspermum ammi essential oil against human bacterial. Int J Adv Biol Biom Res 2014; 2(1): 18-24.
- Gandomi H, Abbaszadeh S, Jebelli Javan A, et al. Chemical constituents, antimicrobial and antioxidative effects of Trachyspermum ammi essential oil. J Food Process Preserv 2014; 38(4): 1690-1695.
- Fazeli-nasab B, Fooladvand Z. A review on Iranian Carum copticum (L.): Composition and biological activities. European J Med Plants 2016; 12(1): 1-8.
- Adams RP. Identification of essential oil components by gas chromatography and mass spectrometry. 3rd Ed. Carol Stream, USA: Allured Publishing Corporation 2001; 455.
- Chandrasekaran M, Venkatesalu V. Antibacterial and antifungal activity of Syzygium jambolanum seeds. J Ethnopharmacol 2004; 91(1): 105-108.
- Mann CM, Markham JL. A new method for determining the minimum inhibitory concentration of essential oils. J Appl Microbiol 1998; 84(4): 538-544.
- Kleinbaum D, Klein M. Survival analysis: A self-learning text. 2nd ed. New York, USA: Springer 2005; 266-286.
- Sikkema J, De Bont JAM, Poolman B. Interactions of cyclic hydrocarbons with biological membranes. J Biol Chem 1994; 269: 8022-8028.
- Denyer SP, Hugo WB. Mechanisms of action of chemical biocides. Oxford, UK: Blackwell Scientific Publication 1991; 171-188.
- Khanzadi S, Gharibzadeh S, Raoufy M, et al. Application of artificial neural networks to predict clostridium botulinum growth as a function of Zataria multiflora essential oil, pH, NaCl and temperature. J Food Safety 2010; 30: 490-505.
- Dorman HJD, Deans SG. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J Applied Microbiol 2000; 88: 308-316.
- Vitali LA, Beghelli D, Biapa Nya PC, et al. Diverse biological effects of the essential oil from Iranian Trachyspermum ammi. Arabian J Chem 2016; 9(6): 775-786.
- Lucchesi ME, Chemat F, Smadja J. An original solvent free microwave extraction of essential oils from spices. Flavour Frag J 2004; 19: 134-138.
- Mohagheghzadeh M, Faridi P, Ghasemi Y. Carum copticum Benth. & Hook. essential oil chemotypes. Food Chem 2007; 100: 1217-1219.
- Mahmoudzadeh M, Hosseini H, Nasrollahzadeh J, et al. Antibacterial activity of Carum copticum essential oil against Escherichia coli O157:H7 in meat: Stx genes expression. Curr Microbiol 2016; 73(2): 265-272.
- Zarshenas MM, Mohammadi Samani S, Petramfar P, et al. Analysis of the essential oil components from different Carum copticum L. samples from Iran. Pharmacognosy Res 2014; 6(1): 62-66.
- Celikel N, Kavas G. Antimicrobial properties of some essential oils against some pathogenic microorganisms. Czech J FoodSci 2008; 26: 174-181.
- Singh A, Singh RK, Bhunia AK, et al. Efficacy of plant essential oils as antimicrobial agents against Listeria monocytogenes in hotdogs. LWT Food Sci Technol 2003; 36: 787-794.
- Tassou C, Koutsoumanis K, Nychas GJE. Inhibition of Salmmonella enteritidis and Staphylococcus aureus in nutrient broth by mint essential oil. Food Res Int 2000; 33: 273-280.
- Carmen Creţu, Floriştean V, Carp-cărare M, et al. The influence of pH and temperature on salmonella spp. from fresh, chilled and frozen poultry carcasses. Cercet agron Mold, XLII, No. 2, 2009; 138.
- Koutsoumanis K, Lambropoulou K, Nychas GJE. A predictive model for the non-thermal inactivation of Salmonella enteritidis in a food model system supplemented with a natural antimicrobial. Int J Food Microbiol 1999; 49: 63-74.
- Theron MM, Lues JF. Organic acids and food preservation. New York, USA: Taylor & Francis 2011; 133.
- Pascual C, Robinson TP, Ocio MJ, et al. The effect of inoculum size and sublethal injury on the ability of Listeria monocytogenes to initiate growth under suboptimal conditions. Lett Appl Microbiol 2001; 33: 357-361.
- Valero A, Rodríguez M, Carrasco E, et al. Studying the growth boundary and subsequent time to growth of pathogenic Escherichia coli serotypes by turbidity measurements. Food Microbiol 2010; 27: 819-828.
- Koutsoumanis KP, Kendall PA, Sofos JN. A comparative study on growth limits of Listeria monocytogenes as affected by temperature, pH and aw when grown in suspension or on a solid surface. Food Microbiol 2004; 21: 415-422.
- Skandamis PN, Stopforth JD, Kendall PA, et al. Modeling the effect of inoculum size and acid adaptation on growth/no growth interface of Escherichia coli O157:H7. Int J Food Microbiol 2007; 120(3): 237-249.
- Gibson AM, Bratchell N, Roberts TA. Predicting microbial growth: Growth responses of salmonellae in a laboratory medium as affected by pH, sodium chloride and storage temperature. Int J Food Microbiol 1988; 6: 155-178.

